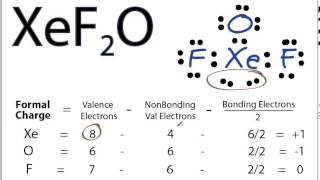 
Use your mouse (computer) or fingers (touch screen) to manipulate the Jmol model. Note that the shapes of the spheroids do not represent the actual electron densities of the lone pairs, but the shapes allow you to visualize how the presence of the lone pairs dictates the molecular geometry. The bond angle between the axial F atoms and the central Cl atom is also compressed from 180° to ~174° for the same reason.Ī cartoon model of the electron densities of the lone pairs of electrons, represented by translucent purple spheroids, can be toggled on and off. The F eq-Cl-F ax bond angles (~87°) are compressed slightly from the values observed in a molecule with both trigonal bipyramidal electron-domain and molecular geometries (90°) due to the lone pairs spreading out more in space than the bonded pairs, compressing the bond angles. The lone pairs of electrons are found in equatorial positions since there is less electrostatic repulsion between the lone pairs and the bonded electron domains than would be present if they were in axial positions. The trigonal -bipyramidal electron-domain geometry. With an expanded octet of ten electrons, a. 1: Common Structures for Molecules and Polyatomic Ions That Consist of a Central Atom Bonded to Two or Three Other Atoms. electron domains, giving a trigonal bipyramidal e-electron-domain geometry and a T-shaped molecular geometry. 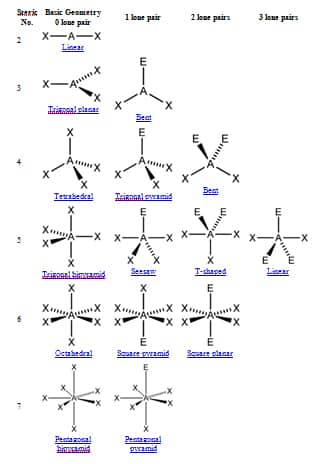
The S atom has five electron domains around it, giving rise to a trigonal -bipyramidal electron -domain geometry. molecular-geometry/ WebWhat is the electron geometry of clf3 Chlorine. The Lewis structure and electron-domain geometry of SF. ClF 3 contains 3 bonded and 2 nonbonded electron domains, giving a trigonal bipyramidal e - electron-domain geometry and a T-shaped molecular geometry. electron-domain geometry and a T-shaped molecular geometry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
